%20Systems-resized-601.png)
Department of Chemistry and Biochemistry, Miami University, Oxford, Ohio 45056, United States
J. Chem. Educ., 2013, 90 (4), pp 500–505
DOI: 10.1021/ed300340x
Publication Date (Web): March 15, 2013
Copyright © 2013 The American Chemical Society and Division of Chemical Education, Inc.
Abstract
The large class sizes of first-year chemistry labs makes it challenging to provide students with hands-on access to instrumentation because the number of students typically far exceeds the number of research-grade instruments available to collect data. Multifunctional chemical analysis (MCA) systems provide a viable alternative for large-scale instruction while supporting a hands-on approach to more advanced instrumentation. This study describes how the capabilities of MCA systems are extended to introduce liquid chromatography (LC) and flow injection analysis (FIA) in undergraduate laboratories. A semi-micro plastic cuvette with a Teflon tubing insert is fashioned as the flow cell for a MCA absorbance–fluorescence detector. Two MCA systems, Vernier and MeasureNet, are used in two unique experiments demonstrating the detection of salicylate in aspirin tablets by FIA and the LC separation of a mixture of riboflavin and fluorescein. Both instruments, composed of a syringe pump, T-injection valve, and the MCA detector, operated in the kinetic mode, are rugged and inexpensive permitting student construction, if desired.
 LUMINESCENCE/PHOSPHORIMETRY
LUMINESCENCE/PHOSPHORIMETRY
Last, but certainly not least, is the use of the MeasureNet “Colorimeter” as a “Phosphorimeter”… also called a “Luminometer”… which, is one of the more versatile Analytical “tools” not readily found in most laboratories. A “Phosphorecent” response can be induced by UV-fluorescence AFTER the light souce is removed… or it can be caused by a chemical reaction that causes the “emission” of VISIBLE Light photons. The proprietary design of the “Colorimeter” optical layout allows the analyst to get the desired response from their sample material… again using the same high-sensitivity optics.
Bio-Luminescence in Fish and Algae, Luciferin/Luciferase Reaction in Fireflies, UV-Blocking capacity of SPF Lotions, the ‘controllable' oxidation of Luminol Light-Sticks, the newly developed photo-luminescent additives for plastics used in skyscraper building and more are beginning labs you can use directly as-is, or modify for your own specific curriculum… as outlined for your review:
Luminescence (“Phosphorescence”) for Kinetic & Relative Comparison values:
• Check out these Glow-in-the-Dark Plastics… some are made of material quite Fantastic!
• Bio-Luminescent Algae and Bacteria are found in “Healthy” Waters… See anything “Fishy”?
• P-AminoBenzoic Acid is the SPF of “Old”… using Luminscence its tale can be told!
• “UV” Light causes “Glow-in-the-Dark” Photon Motions… “Block” it with some SPF Lotion!
• “Luciferase” is the enzyme in the Fire-Fly… catch a few & give this Phosphorimetry Lab a try!
• The Luminol “Light Stick” reaction… uses Organic Oxidation to set those Photons in action!
Many current MeasureNet users that have the MDBC-138 “Colorimeter”… and potential customers, too… may not be aware of the tremendous versatility of this spectral device… and many potential *new* Customers in academia can save a significant amount of time, money and resources by implementing this technology for their teaching laboratories and educational curricula. Designed for simple, direct plug-n-play capability with any MeasureNet Workstation.
QUANTUM FLUORESCENCE & KINETIC SPECTROMETRY
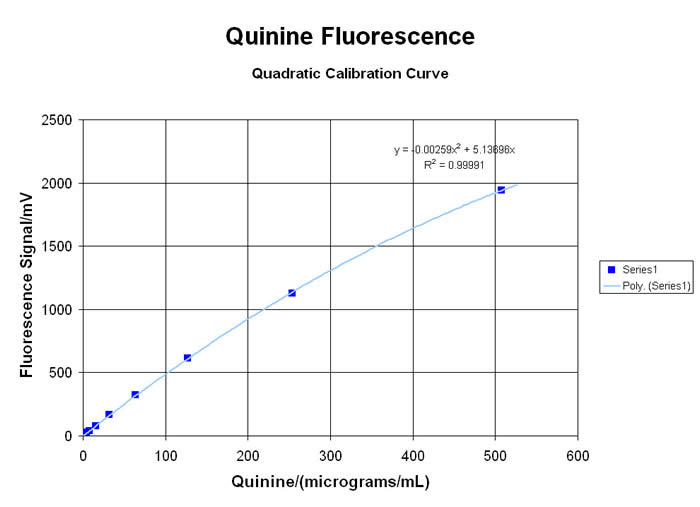
Many types of organic molecules will exhibit a VISIBLE fluorescent “emission” when exposed to high energy ULTRA-VIOLET light to create the “excitation”. This spectro-chemical response is due to the interaction of the energetic UV photons with the loosely-held pi-electrons and other “labile” functional groups in aromatic, olefinic, alkaloidal, xanthinoid and poly-cyclic compounds… and there are many important applications for this little-understood, rarely-taught, under-utilized technology. Using the newest in LED technology, the MeasureNet “Colorimeter” has several “excitation” sources available… with the 375nm UV-LED providing the most useful range of responses for relative fluorescence analyses (since it is very close to the classic 366nm long-wavelength UV from a mercury lamp)… although several alternate user-defined Wavelengths are available from a growing list of LED sources. Please inquire for more information!
When used in the “Fluorometer” mode, the detector is situated at a 90° angle from the UV LED Source, to generate TRUE fluorescent “emission” data without potential interference from the “excitation” wavelength. Laboratory exercises for both qualitative comparisons and quantitative analysis of many materials are possible with the MDBC-138 Dual-Beam Colorimeter. Some applications in Organic Chemistry, Analytical Sciences, Biology/Biochemistry, Environmental and Nutritional Science programs, are highlighted here:
UV-LED Fluorometry for Single-range EMISSION Values:
• “Zap” UV against Chlorophyll from things that Grow… and see what makes it Glow
• The Highlights of Highlighting Markers: How bright is bright?
• Evaluation of Cigarette Second-Hand Smoke: Nicotine for the Masses in your Breathing Gases?
• Analysis of Alkaloid Materials like Quinine
• Measure Amino Acids in “Energy Drinks”… Are their labels Accurate
• Check Ground-Water “Plumes” with Fluoresceine… Water-Table extracts will also be green!
A typical colorimeter is usually a simple, single-beam optical system to measure “color” in the VISIBLE Spectrum of light and provide ABSORBANCE data at a single wavelength. These systems can cost $1,000 or more for stand-alone colorimeters that merely provide simple ABS data… unless you consider the MeasureNet Model MDBC-138 true Double-Beam, Multi-Functional “Colorimeter” based on a unique set of LEDs (Light-Emitting Diodes)... for under $500! The MeasureNet “Colorimeter” is actually FOUR (4) instruments in ONE (1) compact and rugged box… and works as a colorimeter (to make Beer’s Law Curves for VISIBLE Wavelengths), a UV fluorometer (to demonstrate fluorescence & quenching in certain organic molecules), a turbidometer (to measure the turbidity of particulate & colloidal suspensions) and a phosphorimeter (to analyze the phosphorescence [“glow”] of specific materials).
#1) BEER’S LAW ABSORBANCE & KINETIC SPECTROSCOPY
This unit can be used as a “simple” Colorimeter to demonstrate Beer’s Law using three (3) high-output VISIBLE Light LEDs at 472nm (BLUE), 525nm (GREEN) and 630nm (RED); which cover over 75% of the classic General Chemistry and Analytical Chemistry Laboratory experiments that teach spectrophotometric measurements. The wavelength coverage of these LEDs allows highly accurate relative measurement of almost all the ROYGBIV Colors. The disposable & unbreakable 10mm pathlength, near-UV plastic cuvettes included with the “Colorimeter” can be used from ~300nm (in the near-UV) to over 1000nm (in the near-IR) for analytical measurements… and come with sealing caps to preserve prepared solutions for future tests. Some of the popular Laboratory experiments for your Academic Lab curriculum programs Gen Chem, Analytical or Student Research are:
Red / Blue / Green Colorimetric ABS Data for Beer’s Law plots:
• Test for Mineral IRON in your food… check your Cereals, Breads and more
• Unsaturated FATS can be easily seen… just get a purple Color using some IODINE!
• Evaluation of Nutritional Food Proteins… React it to get a “BLUE” and see what can be seen!
• Consistency of M&M and Skittles Candy Colors: Is the "blue" true blue or “faux” to you!
• Changing “Colors” of some pH Dyes… [H+] makes them look *new* to our eyes!
• General Colorimetric Assay… for ANION (X, PO4, SO4, NO3, NO2, etc)
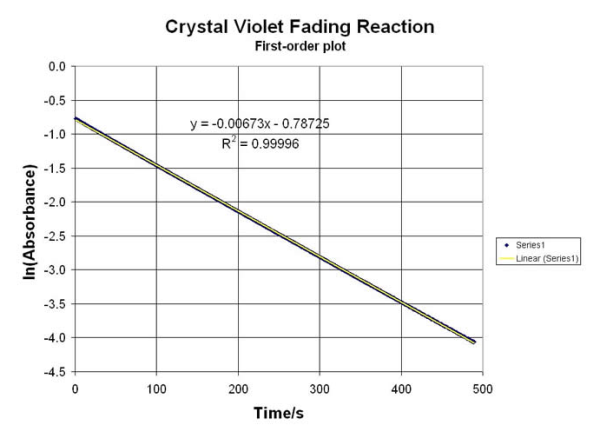
The MDBC-138 is a TRUE Double-Beam Optical System… and provides a reference cell to “blank” out the reagents used to create a very stable ABS reading for several Organic Reactions. Data from these Labs can be used to calculate rate constants, equilibrium factors and reaction conditions (for thermal, ionic and electrochemical variables). This is a MUST for making accurate kinetic experiment tests. A few of them from our “Library” include:
Dual-Beam ABS Data for Kinetic Measurements:
• The Iodine Test for Starches… just Hydrolyze with Amylaze to get a Kinetic Rate
• Perform REDOX “Clock” Reactions
• pH-based Hydrolysis of p-nitrophenylacetate Ester