%20Systems-resized-601.png)
Department of Chemistry and Biochemistry, Miami University, Oxford, Ohio 45056, United States
J. Chem. Educ., 2013, 90 (4), pp 500–505
DOI: 10.1021/ed300340x
Publication Date (Web): March 15, 2013
Copyright © 2013 The American Chemical Society and Division of Chemical Education, Inc.
Abstract
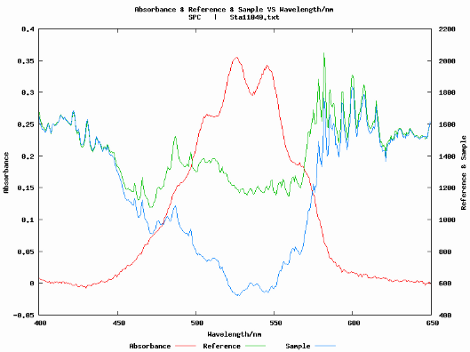
The large class sizes of first-year chemistry labs makes it challenging to provide students with hands-on access to instrumentation because the number of students typically far exceeds the number of research-grade instruments available to collect data. Multifunctional chemical analysis (MCA) systems provide a viable alternative for large-scale instruction while supporting a hands-on approach to more advanced instrumentation. This study describes how the capabilities of MCA systems are extended to introduce liquid chromatography (LC) and flow injection analysis (FIA) in undergraduate laboratories. A semi-micro plastic cuvette with a Teflon tubing insert is fashioned as the flow cell for a MCA absorbance–fluorescence detector. Two MCA systems, Vernier and MeasureNet, are used in two unique experiments demonstrating the detection of salicylate in aspirin tablets by FIA and the LC separation of a mixture of riboflavin and fluorescein. Both instruments, composed of a syringe pump, T-injection valve, and the MCA detector, operated in the kinetic mode, are rugged and inexpensive permitting student construction, if desired.
MeasureNet has added some new features to our LabKonnect™ data storage site and PC software that provide you and your students with even greater flexibility than before.
- Automatic Data Files Graphing
- Online Lab Report Storage/Submission
- Extended Experiments with Remote Monitoring
Automatic Data Files Graphing
With the rollout of the new LabKonnect features, we decided to make it easier to get a quick idea of your data sets. Users can graph any data set uploaded through the MeasureNet Lab PC software right on the LabKonnect™ site.
Online Lab Report Storage/Submission
MeasureNet LabKonnect now allows students to upload their lab reports and store them on the cloud. Instructors can download the lab reports, grade them, and then upload the graded lab report to the student's account for instant feedback with no wasted paper.
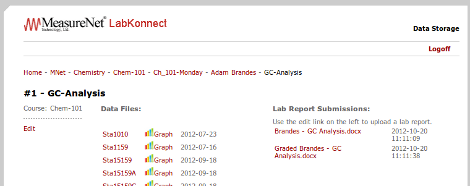
(Lab Report View)
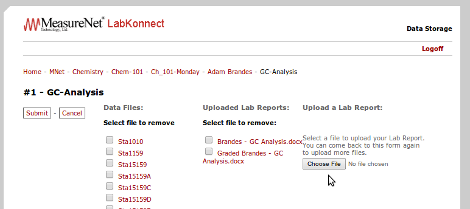
(Lab Report Upload)
Extended Experiments with Remote Monitoring
One of MeasureNet's most exciting new features is the ability to conduct extended length experiments and remotely monitor them from the web. If you have an experiment with a slow reaction that needs to be monitored over several days, you can go home and have fun while still collecting your data. MeasureNet will even send you a text message if your experiment goes out of bounds. That way you can come in and fix your setup without losing days worth of valuable data.
You can set up your alert conditions online, and then start the extended experiment with the Lab PC Software. After that, you just let your experiment run until you choose to stop it.
This capability is ideal for research projects that require the monitoring of experiments that run for days or weeks. The William Heineman research group at the University of Cincinnati has been using this feature for a number of their research projects.
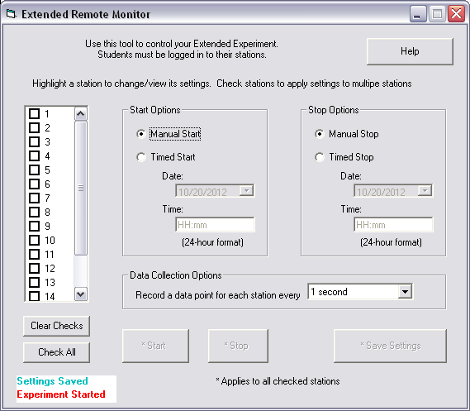
(Setting Up Extended Experiment in Lab PC Software)
If you want to check up on your data and see how it's trending, you can log in and graph or download the data that has been collected so far. You can also leave comments on each of the extended experiment files in case you want to do multiple runs.
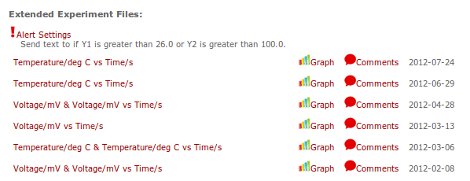
(View and Graph Extended Experiment Files)
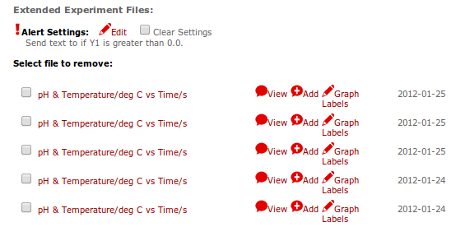
(Mange Extended Experiment Files)
The folks here at MeasureNet have been hard at work integrating new probes into our system and creating new experiments. In this blog entry we'll be introducing the following new experiments and probes:
- Thermometric Titrations
- Conductivity Probe & Experiment Options
- Colorimetric Titration Hardware
Thermometric Titrations
The newest software integrates the drop counter and temperature probe and now has the capability to conduct experiments with Temperature vs Drops. This allows users to conduct Thermometric Titrations.

Conductivity Probe & Experiment Options
MeasureNet now offers a 4 range high resolution conductivity probe. MeasureNet provides users the ability to conduct experiments with Conductivity vs Time and use the drop counter for Conductivity vs Volume for Conductometric Titrations.

Colorimetric Titration Hardware
MeasureNet systems now have the ability to conduct colorimetric, Fluorometric, turbidometric and chemiluminescent titration experiments. When ordering colorimeters, customers can specify if they would like them customized for photometric titrations. The kit includes a colorimeter, a base stand, a pump with power supply, and custom-made flow cell that pair with the MeasureNet colorimeter and drop counter.

The MeasureNet MCAN® (Multi-functional Chemical Analysis Network) system consists of up to fifteen measurement workstations networked together and managed by a single MCAN® Controller and PC. Each Workstation has two +/- 2.5v analog input channels and one high-speed serial communication channel. The analog inputs are sampled by a two channel high-resolution 24-bit Sigma-Delta A/D. Sigma-Delta converters are designed for direct connection to sensors with low signal levels. The built-in signal conditioning and noise reduction of Sigma-Delta data converters makes them ideal for the low level noisy signals often found in potentiometric measurements of high impedance sensors like pH, ISE and other electrochemical sensors. The high-speed serial channel is for sensors with digital outputs.
The MCAN® workstation measurements are displayed in real-time on the workstation LCD and/or streamed in real-time to MeasureNet's LabKonnect cloud server for storage. Data collected at the workstations can be stored locally on the system PC and/or in cloud data storage accounts. Cloud data can also be monitored in real-time from any internet connected device - computer, tablet or smart phone, allowing the researcher to follow the progress from virtually anywhere while running experiments for extended time periods. LabKonnect will also alert the researcher via text message if something has gone wrong. The researcher specifies a range; the system notifies team members if measurements go beyond that range. Researchers no longer need to spend valuable time and resources babysitting experiments.
Comparison of the Effects of Biofouling on Voltammetric and Potentiometric Measurements
Kuhlmann, J., Dzugan, L. C. and Heineman, W. R. (2012), Comparison of the Effects of Biofouling on Voltammetric and Potentiometric Measurements. Electroanalysis, 24: 1732–1738. doi: 10.1002/elan.201200194
Abstract
Biofouling of sensors is a common problem when measuring biological samples. The adherence of proteins and biomolecules, called hemostasis, is the first of four steps that lead to biofouling and eventually a foreign body response. This typically occurs within the first hours after the exposure of the biosensor to a biological sample. The purpose of this study was to assess the effect of this initial step of biofouling on cyclic voltammetry and potentiometric measurements. The results show that biofouling occurred rapidly within minutes and strongly affected cyclic voltammetry measurements, while were minimally affected even after 24 hours.
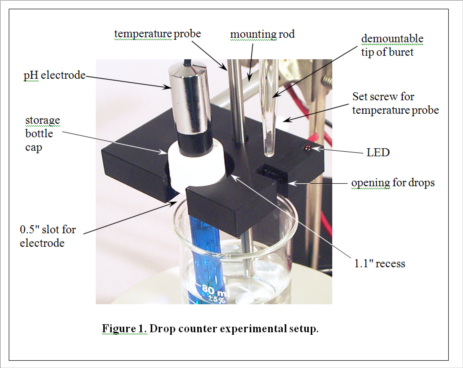
Development of MeasureNet’s optical drop counter began in 2000. Our goal was not only to automate, simplify and shorten the time for pH titration experiments, but also to improve the accuracy of students’ measurements. During its development, various innovative features were added to the design, as seen in Figure 1. MeasureNet’s Multi-Functional Drop Counter holds both a pH probe and a temperature probe, reducing the hardware needed to set up pH titration experiments, and the geometry was optimized to allow the use of small beakers and samples.
Before the days of optical drop counters, simple wire electrical conductivity devices were the only automated and economical solutions for pH titrations in the teaching laboratory. A drop from the buret would make contact with two bare wires positioned closely together, creating a conductive current path. An electronic circuit would then convert this current into a pulse that was counted. A student could then calculate volume based on the number of drops and the average drop size, determined in a separate experiment. The MeasureNet system automatically determines the average drop size in each titration.
The most common method of performing pH titrations is the manual method. The drawbacks to this method:
Time consuming, giving students time to perform few titrations in a lab period.
-
Larger reagent volumes required, making titrations more expensive in terms of reagent usage and disposal.
-
Requires repetition for students to master endpoint detection with reasonable precision.
The 22nd BCCE at Penn State University marks the 10th anniversary of MeasureNet Technology’s introduction of the Optical Drop Counter to the chemical education community. During this decade, the MeasureNet drop counter has become a star in chemistry labs around the country and the world, performing a variety of functions even its designers had never imagined. MeasureNet’s drop counter allows students to leap past the typical pH titration experiment and engage in other interesting and educational titration methods like thermometric, potentiometric, amperometric and colorimetric titrations.
MeasureNet Technology's introduction of the industry’s first Multi-Functional Optical Drop Counter technology in 2002 at the 17th BCCE at Western Washington University obviously caught the attention of the conference attendees and venders. The MeasureNet drop counter has emerged as the industry standard, copied by all its competitors, such as Vernier Software & Technology, Pasco Scientific and MicroLab Inc. MeasureNet is both flattered by this attention and inspired to do more to bring innovative technology into the teaching laboratory.

WVXU Podcast link by Ann Thompson: Focus on Technology: Bugs Cleaning Wastewater
Cincinnati scientists are engineering special bugs that will clean wastewater and create energy. Ann Thompson takes you into the lab where this is happening in Focus on Technology.
By Ann Thompson
MeasureNet Research Applications
MeasureNet is renowned for putting cutting-edge technology into the hands of students but MeasureNet may someday be famous for helping scientists find new and innovative ways for dealing with the world’s most difficult problems.
The technology provided by MeasureNet combines a high-resolution measurement workstation with their LabKonnect cloud-based software to create a system that allows researchers to monitor their experiments from anywhere. Researchers access information from the cloud, important in experiments lasting for a week or longer.
LabKonnect will also alert the researcher via text message if something has gone wrong. The researcher specifies a range; the system notifies team members if data goes beyond that range. Scientists no longer spend valuable time and resources babysitting experiments.
This technology was put through its paces recently, when MeasureNet teamed with Dr. Dan Hassett, who creates special bugs that will clean wastewater and create energy. Hassett, molecular genetics professor at the University of Cincinnati, thinks he has found a way to convert sewage into clean water and energy.
Wastewater has stored up energy in the form of pollutants. Hassett is developing bacterial robots, or “bactobots,” that break down these pollutants and release the energy. Sewage treatment plants become biological fuel cells that produce both clean water and energy.
The bactobots are tiny, only about three microns long, but they generate about 400 milivolts with fluctuations as high as 700 milivolts as they clean the water. Hassett increases the amount of power a bactobot can generate through a series of genetic mutations. Measuring the output of these miniscule bacteria is a big job, and that’s where MeasureNet steps in.
MeasureNet helped Dr. Hassett monitor the voltage and current output of biological fuel cells. Typically, the ouput voltage fluctuates and over the course of four days. Thanks to the sensitive measuring equipment and cloud capabilities provided by MeasureNet technology, Hassett found the existence of a second particular bacterium actually increased output over those four days.
While the output from a single bactobot is small, the impact of these biological fuel cells could break one of humanity’s most vexing vicious cycles. Wastewater treatment plants are the single largest consumer of energy, and the second largest user of water is energy production. Introducing a bacteria that would simultaneously clean water and produce energy would be monumental.
MeasureNet is at the forefront of hands-on laboratory technology, both in the classroom and in the research lab. Like technology itself, MeasureNet continuously develops new ways to enhance the lives and learning of students, scientists and everyday people.

The MCAN® Concept
The MeasureNet Multifunctional Chemical Analysis Network (MCAN® ) is an innovative analytical instrumentation eliminating a multitude of the obstacles that are associated with the PC-based lab systems. With the consolidation of student data acquisition workstations into a solitary network for each group of students, expensive hardware upgrades, computer viruses and the footprint of large equipment is eliminated.
The student workstations we provide interface with numerous kinds of probes as well as other apparatus that are utilized in chemistry laboratories. This allows for a broad range of general chemistry, physical chemistry and biochemistry lab experiments.
MeasureNet can be useful in a freshman chemistry lab or in advanced chemistry labs. Whether used in the freshman chemistry lab, biochemistry, STEM, or environmental chemistry laboratories, MeasureNet is an essential tool.
With MeasureNet, the student takes the measurements at their workstation and the results of those measurements can be stored and then monitored on a single central computer. This allows the instructor to follow student progress and data files. There is no need to contend with the multiple headaches that are associated with managing student lab data because MeasureNet can do it all for you.
Solutions in the Chemistry Lab with MeasureNet
- Less bench space due to energy efficient workstations with smaller foot prints.
- Securely Protected online data storage for students.
- Viruses and computer re-imaging are things of the past with MeasureNet.
- The collection of data electronically allows for more time being spent on the actual experiments.
- Eliminates the need to upgrade large numbers of computers every few years.
- The best quality, research grade chemistry probware is utilized to collect high-resolution data.
General Chemistry Lab Experiments Performed with MeasureNet
MeasureNet was designed to offer instructors considerable academic flexibility with its ability to support a wide range of experiments.
MeasureNet has the ability to support experiments for individual or collaborative student projects in a freshman chemistry lab or in an advanced lab.
The adoption of electronic data acquisition does not require the disposal of experiments that have been proven to build the students’ skills or enhance their understanding of imperative concepts. The longstanding experiments can very easily be integrated into curricula combined with the experiments appropriate for MeasureNet. Other experiments requiring conversion can be modified with the assistance of our curriculum specialists.
Title examples for Guided inquiry, Self-Directed, POGIL, Verification-style and STEM experiments utilizing MeasureNet are as follows:
- Specific Heat of a Metal
- Hot & Cold Packs- Dystan Medical Supply Company
- A Colligative Property of Solutions-Freezing Point
- Quality control at the GlassEX Company-Self-Directed
- Buffer & pH Solutions
- Proteins & Amino Acids
- Chemical Kinetics
- Gas Laws
- Voltaic Cells
- Substances Specific Heat
- Analysis of Metals Emissions
- Heat of Vaporization & Vapor Pressure
- Determining Chromium (VI) Concentrations using Absorption Spectroscopy
- Colligative Properties
- Determining the Cause of a Fish Kill Located in the Clark Fork of the Columbia River
- Identification of a Weak Unknown Acid
- Analysis of the Phosphorus in Cola
- Identification of an Unknown Metal-Self Directed
- Determining the Ka Value of a Weak Acid
- Determining the concentration of Acetic Acid in Vinegar
- Determining the Molecular Weight, with the use of the Ideal Gas Law, of a Volatile liquid
- Hess’ Law-Enthalpy of Reaction
- Analysis of the Phosphorus in Fertilizer
- Determining the Heat of Neutralization for Various Strong Bases & Acid
- Reaction Stoichiometry & Moles
- Determining a Reaction Equilibrium Constant with the Use of Absorption Spectroscopy
- Analysis of Emission of Aequeous Solutions from Group IA & IIA Metal Salts
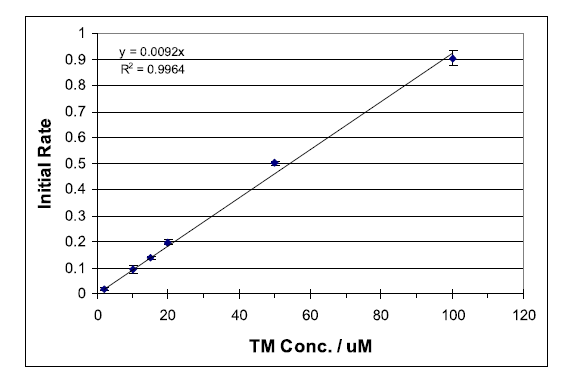
Measurement of molecular fluorescence is an important analytical technique in chemical and biological sciences. The capacity of detecting very small fluorophore concentrations, or small changes in its concentration, combined with high specificity make this technique a very powerful analytical tool.
Kinetic methods for determining reaction rates are commonly used. While most experiments are designed to determine the order of a particular reaction in order to gain insight to the reaction mechanism, kinetic methods are also used for quantitative analysis. Determination of the initial reaction rate is one way of quantitatively analyzing a compound within a suitable reaction system.
The following experiment is designed to introduce students to both of these concepts, fluorescence and kinetics as analytical method. This combined technique has the advantage of increased analyte specificity over equilibrium-based fluorescence measurements. As only the compound that is reacting is causing a change in the measured fluorescence signal, steady-state interferences are largely eliminated.
Thiamine (vitamin B1) is essential for the metabolism of carbohydrates and normal function of he nervous and cardiovascular systems. Severe vitamin B1 deficiency will eventually lead to beriberi, characterized by abnormal functions of the muscular and nervous systems, as well as heart and brain abnormalities. Vitamin B1 occurs naturally in foods like whole grains, nuts, vegetables, pork, and liver.
Thiamine (TM), a non-fluorescent compound, has been found to be oxidized selectively by mercuric oxide (HgO) at a rate suitable for monitoring with standard fluorescence spectrometers.1 The oxidation product, thiochrome (TC), is fluorescent with a strong absorbance maximum at 367 nm and fluorescence emission at 444 nm. Deprotonation of TM yields a non-fluorescent tricyclic intermediate (CI), which is oxidized to TC as outlined in Scheme 1. Immediate oxidation of CI is essential to avoid the formation of several non-fluorescent productions.1 Thus, it is important to follow the sequential addition of reactants as outlined below... To view the complete experiment Click Here.
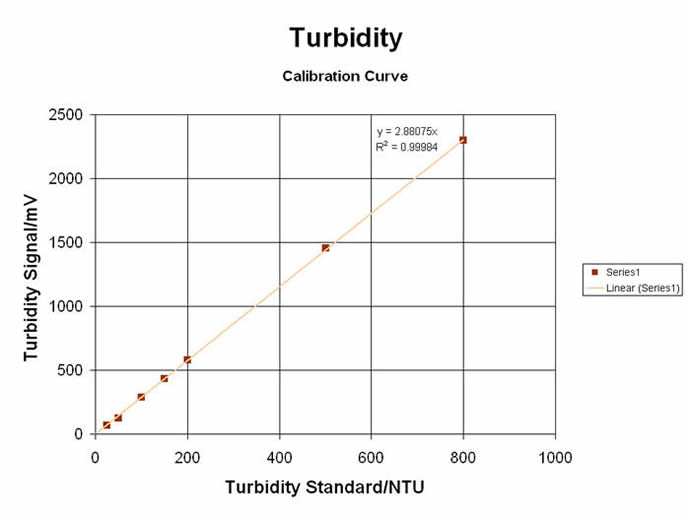
#3) TURBIDIMETRIC / NEPHELOMETRIC LIGHT-SCATTERING SPECTROMETRY
The “Colorimeter” can also be used in the “Turbidometer” mode… or more precisely, as a “Nephelometer”, since nephelometry uses a Near-IR LED (@ 880nm) to measure the SCATTERED light at a 90° angle from the light source, as it is done on the MeasureNet unit. Tests can be made on sample solutions that have existing particulate suspensions… or a reaction can be made to produce a range of particulates for relative and direct measurements. Originally used for Clinical Chemistry applications, Turbidometry/Nephelometry can be applied to a wide range of curriculum subjects… from Organic Synthesis & Analytical Chemistry to Environmental & Biochemistry Labs. Some of these unique and educational labs are shown here:
Turbidity (“Nephelometry”) for Kinetic & Relative Comparison at Single values:
• Environmental Testing of “Settleable Solids” for River / Stream & Estuarial Waters
• Reducing Sugars by Benedict’s Test... great for Diabetics, Atkins-Diets and all the rest!
• Collect some of the DUST from your “Air”… is it okay to breathe or is something there?
• Rate of Reaction of Silver Ion + Chloride Ion to form AgCl precipitate… Kool Kinetics!
• The classic Princeton “Nassau Clock” Reaction… makes for a great teaching Lab attraction!
• Measurement of Yeast Growth in Beer… is it fermenting slow or in high-gear?
QUANTUM FLUORESCENCE & KINETIC SPECTROMETRY
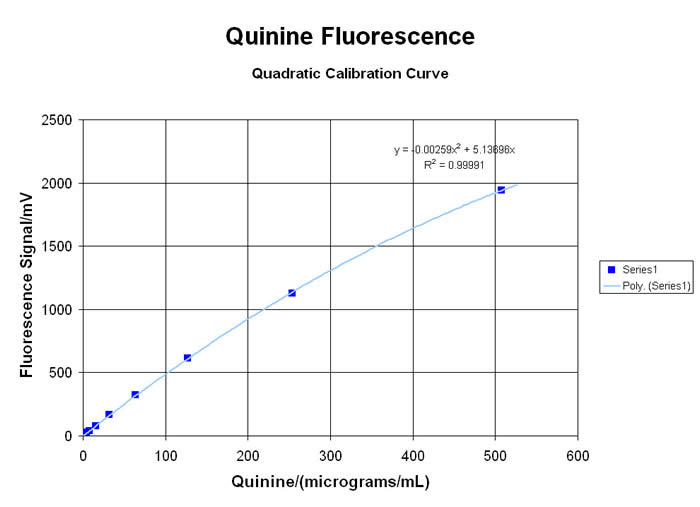
Many types of organic molecules will exhibit a VISIBLE fluorescent “emission” when exposed to high energy ULTRA-VIOLET light to create the “excitation”. This spectro-chemical response is due to the interaction of the energetic UV photons with the loosely-held pi-electrons and other “labile” functional groups in aromatic, olefinic, alkaloidal, xanthinoid and poly-cyclic compounds… and there are many important applications for this little-understood, rarely-taught, under-utilized technology. Using the newest in LED technology, the MeasureNet “Colorimeter” has several “excitation” sources available… with the 375nm UV-LED providing the most useful range of responses for relative fluorescence analyses (since it is very close to the classic 366nm long-wavelength UV from a mercury lamp)… although several alternate user-defined Wavelengths are available from a growing list of LED sources. Please inquire for more information!
When used in the “Fluorometer” mode, the detector is situated at a 90° angle from the UV LED Source, to generate TRUE fluorescent “emission” data without potential interference from the “excitation” wavelength. Laboratory exercises for both qualitative comparisons and quantitative analysis of many materials are possible with the MDBC-138 Dual-Beam Colorimeter. Some applications in Organic Chemistry, Analytical Sciences, Biology/Biochemistry, Environmental and Nutritional Science programs, are highlighted here:
UV-LED Fluorometry for Single-range EMISSION Values:
• “Zap” UV against Chlorophyll from things that Grow… and see what makes it Glow
• The Highlights of Highlighting Markers: How bright is bright?
• Evaluation of Cigarette Second-Hand Smoke: Nicotine for the Masses in your Breathing Gases?
• Analysis of Alkaloid Materials like Quinine
• Measure Amino Acids in “Energy Drinks”… Are their labels Accurate
• Check Ground-Water “Plumes” with Fluoresceine… Water-Table extracts will also be green!